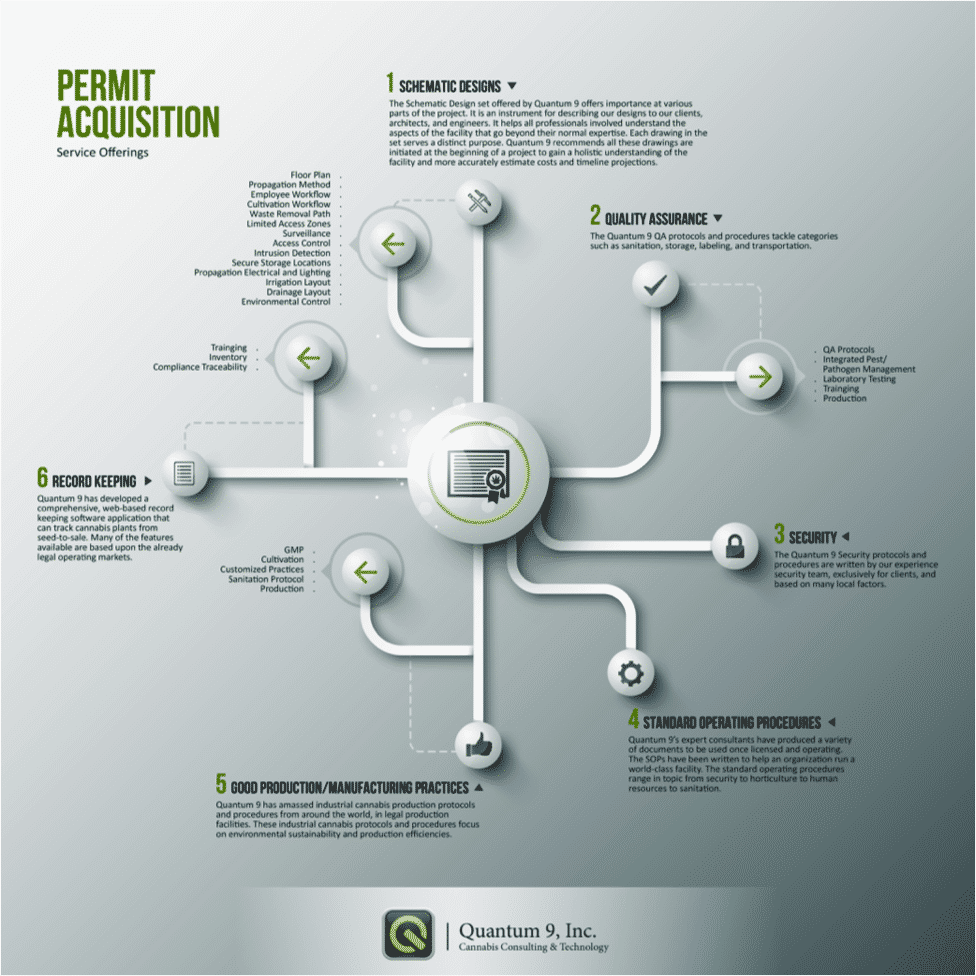
Our license acquisition services are geared to help you get licensed in the medical or adult-use cannabis industry. We start by provisioning a full set of schematic plans for the facility. The facility design process is an integral part of the services offered by Quantum 9, Inc. because the design is the single most important factor in the determination of projected production and subsequent revenue. In addition, the facility design will determine factors such as operational efficiency, cultivation technique, technology integration, security platform, and product purity. Included in the schematic design are the floor plans, propagation method, employee workflow, waste removal path. As well as cultivation workflow, surveillance and security documentation, electrical plans, and many others.
Our technical documentation services include a customized submission application for licensing in your respective market. Quantum 9’s experts will customize, analyze and approve your schematic designs. We will write technical documentation that is exclusively written for the use of our clients. Our expert consultants have produced extensive documentation utilized for license acquisition as well as actual operations. We offer a set of standard operation procedures (SOPs) for every action in your business protocol. Along with the application, the entirety of the set spans over 187 documents and over 2,000 pages. We write the SOPs to help an organization run a world-class facility.
Regulatory bodies require an unprecedented amount of information around quality assurance, good manufacturing processes based on environmental sustainability and production efficiencies. If you are interested in learning more about how the business analysts and consultants at Quantum 9, Inc. can help your project get approved contact us, or read more about service offerings in our blog.
Error: Contact form not found.








Sorry, but your message did not come through completely, would you mind resending it or emailing it to Kevin.Mahoney@quantum9.net. Thank you in advance, Kevin Mahoney.